odour control
Reading time:However, before examining the treatment of gaseous effluent, consideration has to be given to action required in respect of structures, even upstream drains, in order to limit odour emissions (see gaseous, odorous pollutants in a wastewater treatment plant).
There are three potential types of treatment that can be used to control airborne odour in wastewater treatment plants:
- treatment by physico-chemical absorption or scrubbing;
- biological treatments;
- odour control through absorption over activated carbon.
Depending on the target objectives, odour-controlled equipment and structures and constraints specific to the site, these processes can be combined to advantage.
Other, less traditional processes can also be used:
- thermal oxidation;
- photocatalysis based on the principle of oxidation using a catalyst;
- cold plasma.
physical-chemical treatment in scrubbing towers: Azurair™ C
reagents used
Gas is scrubbed in a packing tower, transferring pollution to the liquid phase; this is followed by a chemical reaction: acid-base reaction in the case of acid and base towers, oxidation-reduction reaction in the case of “bleach” (NaCℓO), thiosulphate and bisulphite towers.
Depending on the pollution mixture to be treated, one to four reagents will be needed for its removal. Accordingly, their use in the towers follows a sequence.
A sulphuric acid (H2SO4) scrubbing removes nitrogen compounds and especially ammonia and amines:

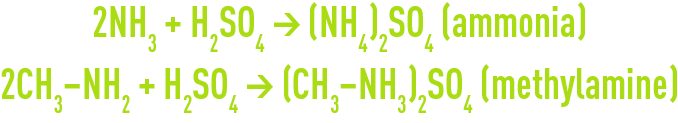
An oxidising scrubbing using sodium hypochlorite (NaCℓO) removed reduced sulphur compounds, especially hydrogen sulphide, organic sulphides, mercaptans and also ammonia and amines:

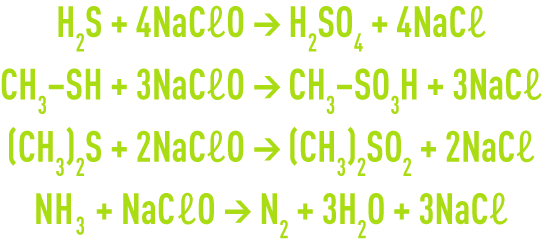
The alkaline scrubbing using sodium hydroxide (NaOH) fixes volatile fatty acids ( VFA ), reduced sulphur and residual chlorine. In particular, sodium hydroxide (NaOH) removes carboxylic acids, hydrogen sulphide and mercaptans as well as a proportion of the carbon (CO2) found in air and this has to be taken into consideration when calculating reagent consumption:

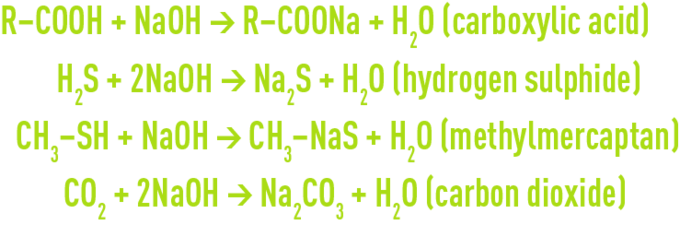
Reduction scrubbing using bisulphate (NaHSO3) or thiosulphate (Na2S2O3) is mainly used to eliminate residual chlorine, as well as, with bisulphate, some aldehydes.
Oxydising and alkaline scrubbers can be used together but to the detriment of the quantities of reagents used.
The above reactions will destroy compounds generating unpleasant odours (e.g. H2S via C`O–) or, more frequently, fix them in the circulating solution which will then have to be drained from time to time and returned to the plant inlet.
schematic diagram of an odour control tower (figure 2)

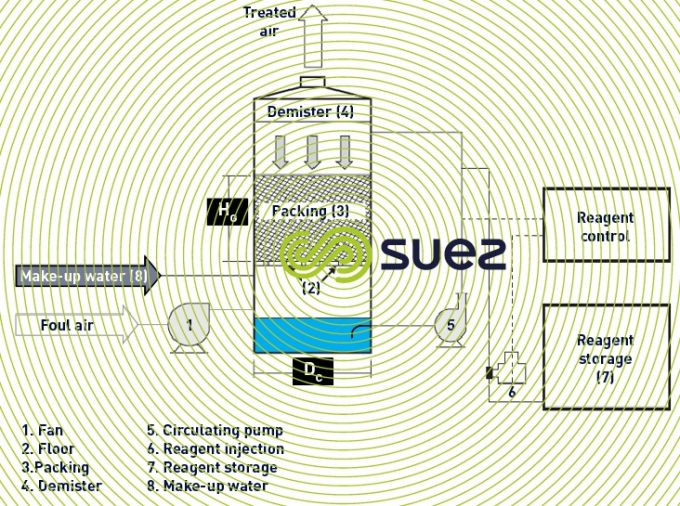

air circulation (figure 2)
A fan (1) is used to send the foul air extracted from a confined building (or structure) into one or more towers in series. The air is injected into the tower beneath the floor (2) supporting the packing (3) through which the reagent percolates. The gaseous compounds are absorbed and the chemical reactions described earlier take place within this packing.
As the air leaves the towers, it goes through a demister (4) that limits the droplets carried, especially from one tower to the next.
circulation of the liquid solution (figure 2)
The solution is drawn up by a centrifugal pump (5) at the bottom of the tower, from a storage capacity, and delivered (QL) to a distribution system (ramp or sprayer nozzles) positioned above the packing.
The circulation flow is set between two limits according to the air flow to be treated: minimum ensuring that the entire packing surface area is wetted and maximum to avoid excess flow. Therefore, these values will depend on the packing selected.
reagent injection-control
The scrubbing solution’s reagent concentration is measured by:
- using a pH metre in the case of an acid, sodium hydroxide, bleach/sodium hydroxide and thiosulphates tower;
- measuring the available chlorine concentration in the case of a bleach and bleach/sodium hydroxide tower;
- measuring the redox potential in the case of a thiosulphate or biosulphate tower.
This concentration is kept to within two limit values by automatic in-line injection (6) of a reagent drawn from a storage capacity (7) (figure 2).
make-up water
Make-up water (8) (QE) is required to at least compensate for evaporation and water carried away in the form of aerosols, despite the inclusion of a demister (4).
The scrubbing solution must be renewed periodically - at least once a week - to limit the concentration of salts. Once yearly, the storage capacity has to be emptied for cleaning and usual inspection procedures.
When the TH is greater than 5°F, make-up water needs to be softened for towers with basic pH.
designing parameters
The treatment system is selected as detailed above, according to the nature and quantity of pollutants to be processed and to target discharge concentration levels. When designing the towers, the following also needs to be allowed for:
- type of packing selected (supplied in bulk or in an orderly sequence, specific surface area …);
- gas velocity through the towers (maximum 2.1 m3TPN·m–2·s–1 in the case of bulk packing), whence the tower’s diameter;
- the liquid solution flow rate (standard); 2.5 L·m–3TPN of air in the case of bulk packing).
The “stringent” guarantee conditions often demanded have led us to regularly put forward a system combining four towers in series: “acid, bleach, bleach/sodium hydroxide, sodium thiosulphate”.
treatment performances
The design and combination of different chemical scrubbing towers are defined according to the required treatment performances. As an indication, table 6 below sets out the performances of traditional odour control by four-tower chemical scrubbing (acid, bleach, soda, sodium thiosulphate).



Note: These performances generally control the olfactory impact of the plant. Where specific constraints exist, higher treatment performances can be achieved by this process by adjusting the design parameters.
activated carbon treatment: Azurair™ A
This process is recommended for low gas flow rate systems with low pollution loading. Consideration can also be given to its use as a polishing stage after biological treatment or, if applicable, after a gas scrubbing.
The treatment principle consists in bringing the air to be purified into contact with a mass of granular activated carbon ( GAC ) or impregnated alumina over which the pollution compounds are adsorbed.
An adsorption filter works satisfactorily within an air temperature range of 5 to 40°C.
The main parameter is the passage speed: 500 m/h if the compounds can only be adsorbed with difficulty to 2000 m/h for the others. To avoid creating too great a head loss, the carbon is about 1 meter deep and the contact time is therefore between 2 and 7 second.
The adsorption capacity depends on the substance(s) adsorbed. When there is a mix of gaseous compounds (as in wastewater treatment plants) according to the affinity of the compounds with the activated carbon, elution phenomena may occur. The air needs to be heated or even dehumidified when it contains water particles (mist) or when there is a danger of condensation.
The performances of the adsorption over activated carbon or impregnated alumina, before saturation of the media, are excellent:
- approximately 99% efficient H2S elimination (for concentrations above 5 mg·m–3);
- possibility of treating punctual H2S peaks to a concentration in the order of 100 mg m–3;
- wide spectrum of adsorbed compounds, especially VOC (volatile organic compounds) of which some are difficult to reduce using other means.
Consideration can be given to a “biflow” design (figure 3) that restricts the system’s footprint.



biological treatments: Azurair™ B
Fixed bacteria are used to purify the air; therefore, this is a biological filter and in the Azurair™ B, support material is a biolite which, however, is not submerged as is the case for a Biofor.
The Azurair™ B with its mineral filtering medium is therefore classed as ‘biotrickling’.
The following schematic diagram applies (figure 4):

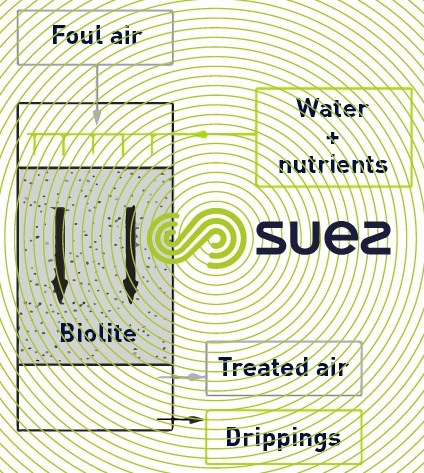

In this instance, the air to be treated circulates down flow at the rate of 400 to 1,200 m3 air·m–2·h–1 depending on the incoming concentration and on the target residual concentration; water is sprayed on the support (600 litres per m3 of biolite and per day). It is used to keep the bacteria film wet and to provide it with the necessary nutrients.
The bacteria oxidise the biodegradable pollutants and the hydrogen sulphide converts into sulphuric acid and the ammonia into nitrate. The pH of dripping can drop down to 1.5 depending on the hydrogen sulphide content and, therefore, block other biological reactions. At this pH , a thiooxidising biomass always degrades the hydrogen sulphide; the other biological reactions are inhibited, however. Drips are returned to the plant inlet.
As in the case of all biological treatment, this process is temperature-sensitive and starts up progressively. The following minimum temperatures apply:
- minimum air temperature of 10 °C;
- minimum spray water temperature of 12 °C.
A wash may be required depending on the loading eliminated and, therefore, on bacterial growth. The wash will be actuated by the head loss.
However, in the case of a properly managed installation, if the pollution load is not too high and if it does not contain any source of carbonated pollution, the wash frequency will be low (1 wash every 2 to 5 years) and, in such cases, a manual wash system will be sufficient.
Less compact and with limited efficiency compared with chemical scrubbing systems, this type of odour control is normally used on small to medium-size plants or on heavily loaded air as part of an odour “pre-treatment” (upstream from an activated carbon or chemical scrub treatment). It can then be used either to comply with highly stringent discharge standards or more often to reduce the consumption of reagents (acid, bleach, sodium hydroxide) and activated carbon.
It should be noted that there are many filters using coarser supports such as peat, compost, bark. However, these need to be calculated for use with gas velocity that are 3 to 5 times lower than for the Azurair™ B. In addition, these beds require regular monitoring to prevent their clogging and that is why we prefer the Azurair™ B.
An example of treatment performances obtained by this process is presented in the table below.



other odour control processes
thermal oxidation
Thermal oxidation is the process whereby air pollutants are oxidised under high temperatures (higher than 800°C). The heat released by the combustion is recovered for external use (recovery oxidiser) or is recovered exclusively to reheat the stale air to be treated (RTO regenerative oxidiser).

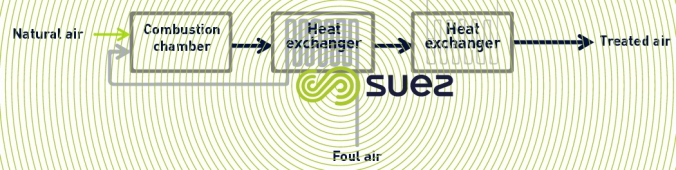


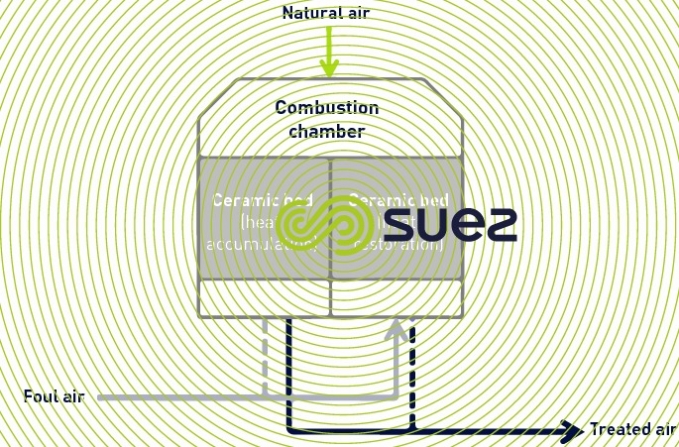

This process is suitable for treating stale air that is highly loaded, mainly with VOC (Volatile Organic Compounds) that chemical scrubbing finds difficult to eliminate. It is very effective (about 98%) in treating odours and VOC . Depending on the characteristics of the gas to be treated, it can be necessary to install an upstream water scrubber to eliminate the nitrogen compounds which oxidise into NOx in the oxidiser.
photocatalysis
The principle of photocatalysis is to create free-radical and ionised species in the air to be treated that have the effect of oxidising the gaseous pollutants. The excited species are created under the effect of UV and a catalyst (normally TiO2).
The UV source can be the sun, which limits the OPEX costs of this technique. In this case, it is important to combine photocatalysis with adsorption over activated carbon to ensure continuous treatment of gaseous pollutants during the night.

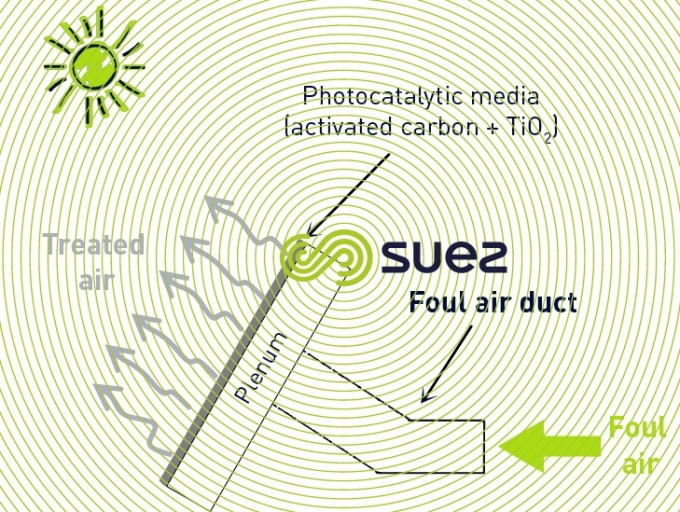

Photocatalysis can control odours in air with low pollutant concentrations. It is applied to the treatment of extracted air in a building atmosphere.
Note: the cold plasma technique operates on an identical principle to photocatalysis: oxidation of gaseous pollutants by free-radical and ionised species contained in the plasma gas. Conversely, the generation of the plasma gas requires a high-voltage generator and therefore results in high electricity consumption.
solutions for reducing the olfactory impact
Encouraging odorous emissions to disperse into the atmosphere is another excellent way of reducing the olfactory impact of an odorous source.
This means discharging the stale air or odour control output through a sufficiently high chimney (more than 10 meters), with a diameter calculated to ensure an ejection speed in the air of around 12 m/s.
Another solution is to install an Azurair™ D. The principle here is to propel the gaseous discharges into the atmosphere at high speed using a pulser like Eolage® from Delamet Environnement.



This is an especially effective technique when meteorological conditions are not favourable to atmospheric dispersion. This technique can be usefully combined with an odour treatment unit with zero nuisance as the goal.
notion of treatment system
The various odour treatment techniques can be combined to optimise their cost/effectiveness operation.
Some examples:
- in wastewater treatment plants dealing with very septic water, the Azurair™ Boost combining biological pre-treatment with an Azurair™ C can eliminate a large proportion of the H2S upstream. The product is then optimised for the consumption of chemical reagents;
- a finish treatment is necessary when there are harsh odour treatment requirements or when the plant is installed in the immediate vicinity of local residents;
- for sludge heat treatments (drying, hydrolysis, carbonisation, wet oxidation), which release odorous VOC , special attention should be paid to treat these flows upstream in particular.
Bookmark tool
Click on the bookmark tool, highlight the last read paragraph to continue your reading later












