general description of the subcritical wet oxidation method
Reading time:This oxidation takes place in reactors that can operate at a temperature of between 220 and 320°C and at a pressure of between 40 and 110 bar. The working pressure is, in fact, kept at a level slightly above the water vapor pressure; under these conditions, the reaction takes place in the liquid phase.
Depending on temperature conditions, oxidation may or may not use a catalyst and the reaction will take between 30 to 120 minutes. In order to ensure that the reaction is self-sufficient in terms of heat, the amount of COD injected into the reactor must be greater than 25 g·L–1 of COD, and the sludge may have to be thickened beforehand.
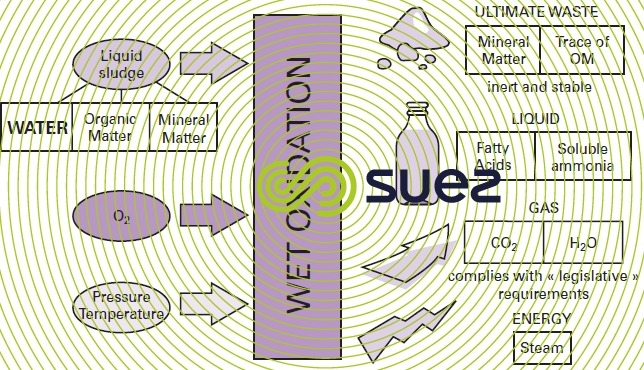
Therefore, wet oxidation is used on sludge that has been concentrated but not dewatered (figure 40), producing the following results :
- 80% of OM will be mineralised as CO2 and H2O, and the remaining 20% solubilised as an organic compound having a simple and easily biodegradable structure (mainly acetic acid);
- sulphur (S), phosphorous (P), chlorine (C`) and nitrogen (N) contained in sludge organic matter will be converted into SO4, PO43–, Cℓ–and NH4+ ions respectively.
Therefore, this system creates :
- Ua gas that mainly consists of CO2 and water vapor; this gas will also contain NH3, CO and VOC (volatile organic compounds); this is a dust-free gas that only contains traces of SO2, NOx, dioxins and furanes. It is processed by catalytic oxidation in order to remove CO and VOC before being discharged into the atmosphere.
- A liquid: the oxidised liquid mainly contains suspended solids, soluble mineral and organic compounds and ammonia produced when the organic nitrogen is oxidised. This liquor then has to be clarified through sedimentation, enabling the mineral matter to be collected before dewatering, without any treatment, from a filter press. This cake only comprises mineral matter including heavy metals found in a stabilised and non-leachingform that can, therefore, be extracted (ultimate waste). This waste may contain a very low percentage of organic matter that decreases as the temperature rises.


Bookmark tool
Click on the bookmark tool, highlight the last read paragraph to continue your reading later












